|
Youll find more specific groups, like transition metals, rare earths, alkali metals, alkaline earth, halogens, and noble gasses. There are multiple ways of grouping the elements, but they are commonly divided into metals, semimetals (metalloids), and nonmetals. All of the elements in a group share the same number of. This is what is meant by periodicity or periodic table trends. Alkali metals typically take on an oxidation state of +1 and prefer to lose an electron to achieve a noble gas-like configuration they and the alkaline earth metals are the most reactive of all metals.Refresher: The periodic table is organized into groups - where each column comprises a group. Mercury metal can be absorbed through your skin, but organic mercury is a much more common threat. Conclusion - The Periodic Table - Review. They decrease in reactivity down the group but increase in their melting and boiling points. H2S represents no considerable reactivity.Ī list of Reactive and Non-Reactive Gases to help when choosing Calibration or Bump test gas cylinders and accessories. The Halogens in Group 17 are non metal elements that are diatomic and simple covalently bonded. Unlike non-reactive gases, reactive gases require higher flow (they will use a 1.0 LPM regulator, instead of a 0.5 LPM) and need corrosion-resistant equipment as corrosive and sticky (stainless steel regulators and Teflon tubing). They get more reactive the further down on the periodic table you go too, with cesium and francium being so reactive that they can burst into flames simply by being exposed to the air. They have the same number of electrons in their outer shell. The most reactive nonmetals reside in the upper right portion of the periodic table. Why are the elements fluorine, chlorine and iodine placed in the same group of the Periodic Table They all readily react with oxygen. Noble gases are the least reactive chemical elements. Alkali metals react vigorously to water and air.These elements will dance around, sizzling due to the production of hydrogen gas, and often explode. You will get a different set of questions each time you attempt this quiz. 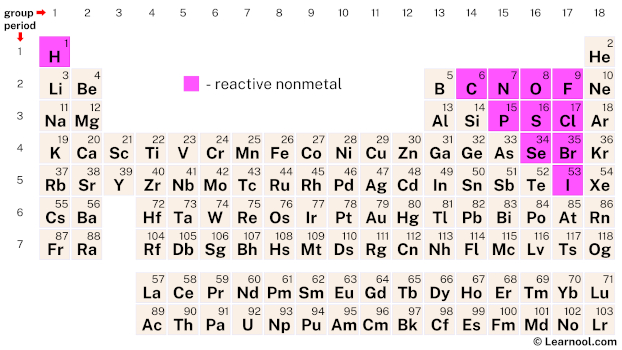
There are seven noble gas elements: helium, neon, argon, krypton, xenon, radon, and oganesson. The principal danger is that failure to use compatible materials and proper calibration procedures can result in dangerously inaccurate (low) readings and increased response times. The noble gases are group 18 on the periodic table, which is the column of elements on the right side of the table. Because of their greater tendency to be depleted from a gas sample by the exposed surfaces of gas detection systems, special care must be taken to ensure accurate monitoring results. As expected, semimetals exhibit properties intermediate between metals and nonmetals.Reactive gases are defined as gases which, because of their high chemical activity, are easily sorbed (adsorbed) by the exposed surfaces of gas detection systems including detector housings, calibration adapters, and remote sample draw accessories (tubing). Most solid nonmetals are brittle, so they break into small pieces when hit with a hammer or pulled into a wire. Nonmetals can be gases (such as chlorine), liquids (such as bromine), or solids (such as iodine) at room temperature and pressure. Nonmetals, in contrast, are generally poor conductors of heat and electricity and are not lustrous. Of the metals, only mercury is a liquid at room temperature and pressure all the rest are solids. Non-metallic tendency increases going from left to right across the periodic table. H2S represents no considerable reactivity. Metallic tendency increases going down a group. Unlike non-reactive gases, these gases require higher flow (they will use a 1.0 LPM regulator, instead of a 0.5 LPM) and corrosion resistant equipment as they are corrosive and sticky (stainless steel regulators and Teflon tubing). 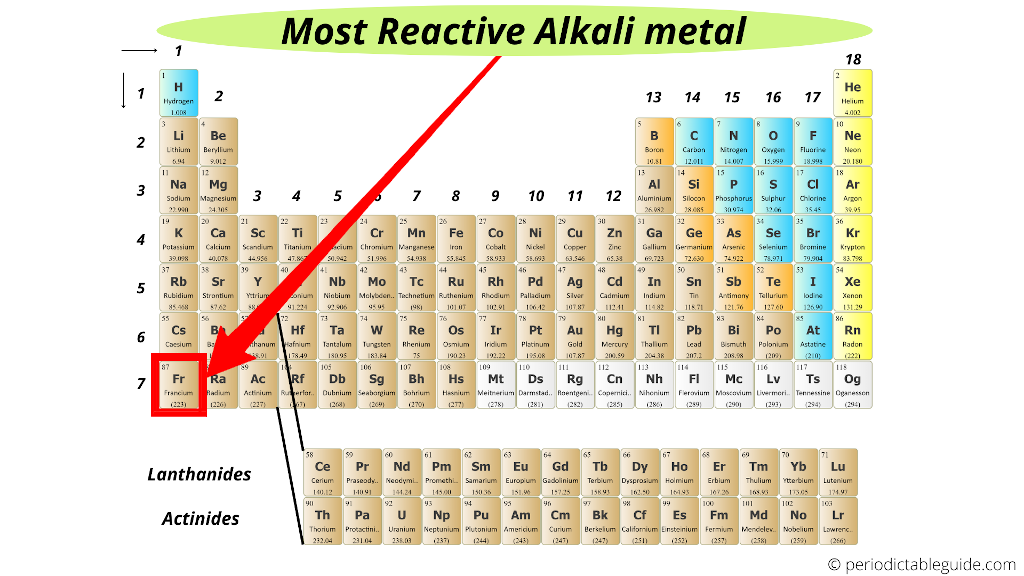
Non-metallic character relates to the tendency to accept electrons during chemical reactions. For example, rubidium has 5 electron shells, a property found in all other period 5 elements, whereas its electron configuration s ending is similar to all other alkali metals: s 1. The vast majority of the known elements are metals. Metallic character refers to the level of reactivity of a metal. It is an alkali metal, the most reactive group in the periodic table, having properties and similarities with both other alkali metals and other period 5 elements. Metals-such as copper or gold-are good conductors of electricity and heat they can be pulled into wires because they are ductile they can be hammered or pressed into thin sheets or foils because they are malleable and most have a shiny appearance, so they are lustrous. 
The distinction between metals and nonmetals is one of the most fundamental in chemistry. Gold-colored lements that lie along the diagonal line exhibit properties intermediate between metals and nonmetals they are called semimetals. \) divides the elements into metals (in blue, below and to the left of the line) and nonmetals (in bronze, above and to the right of the line).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
